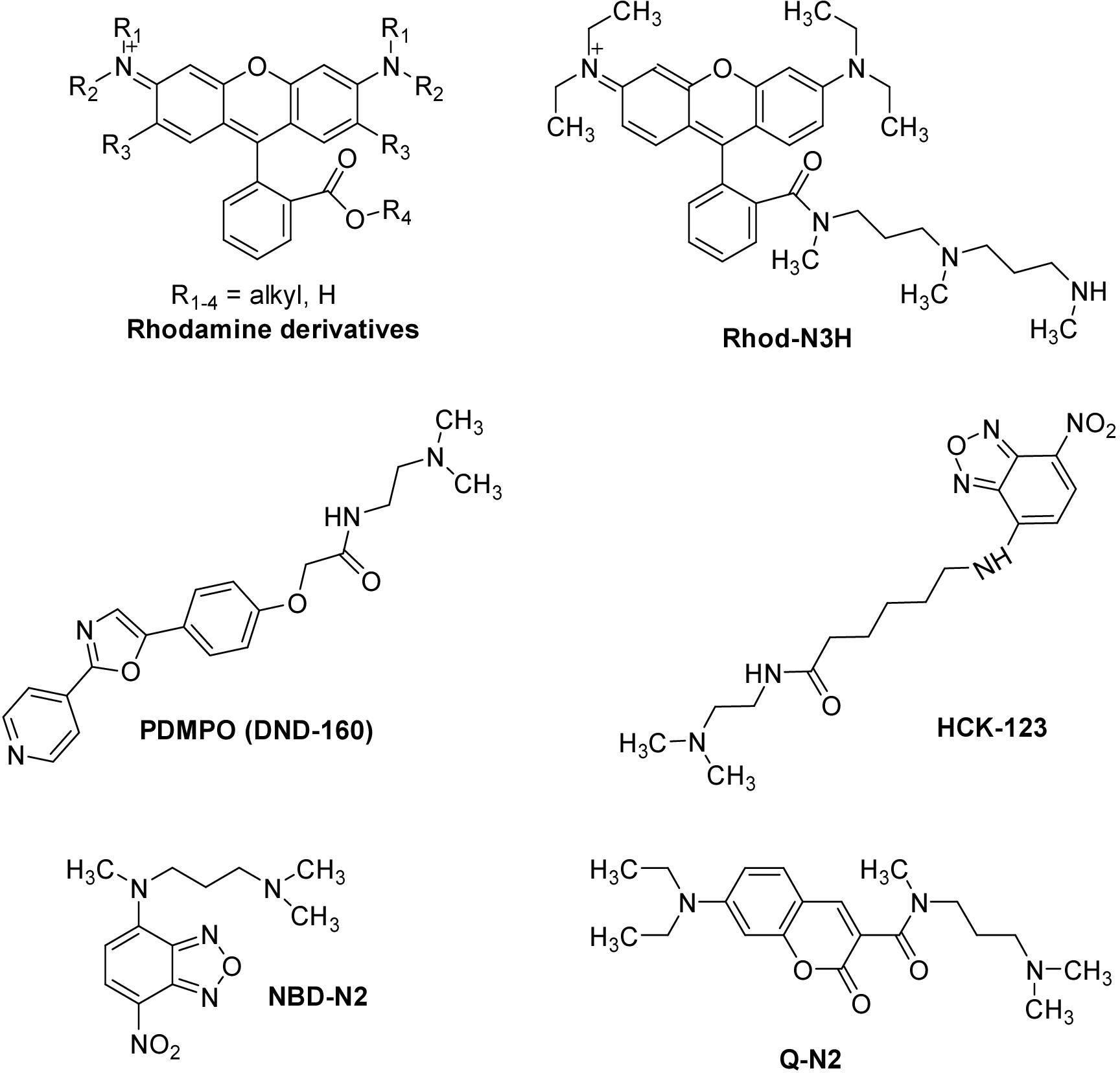
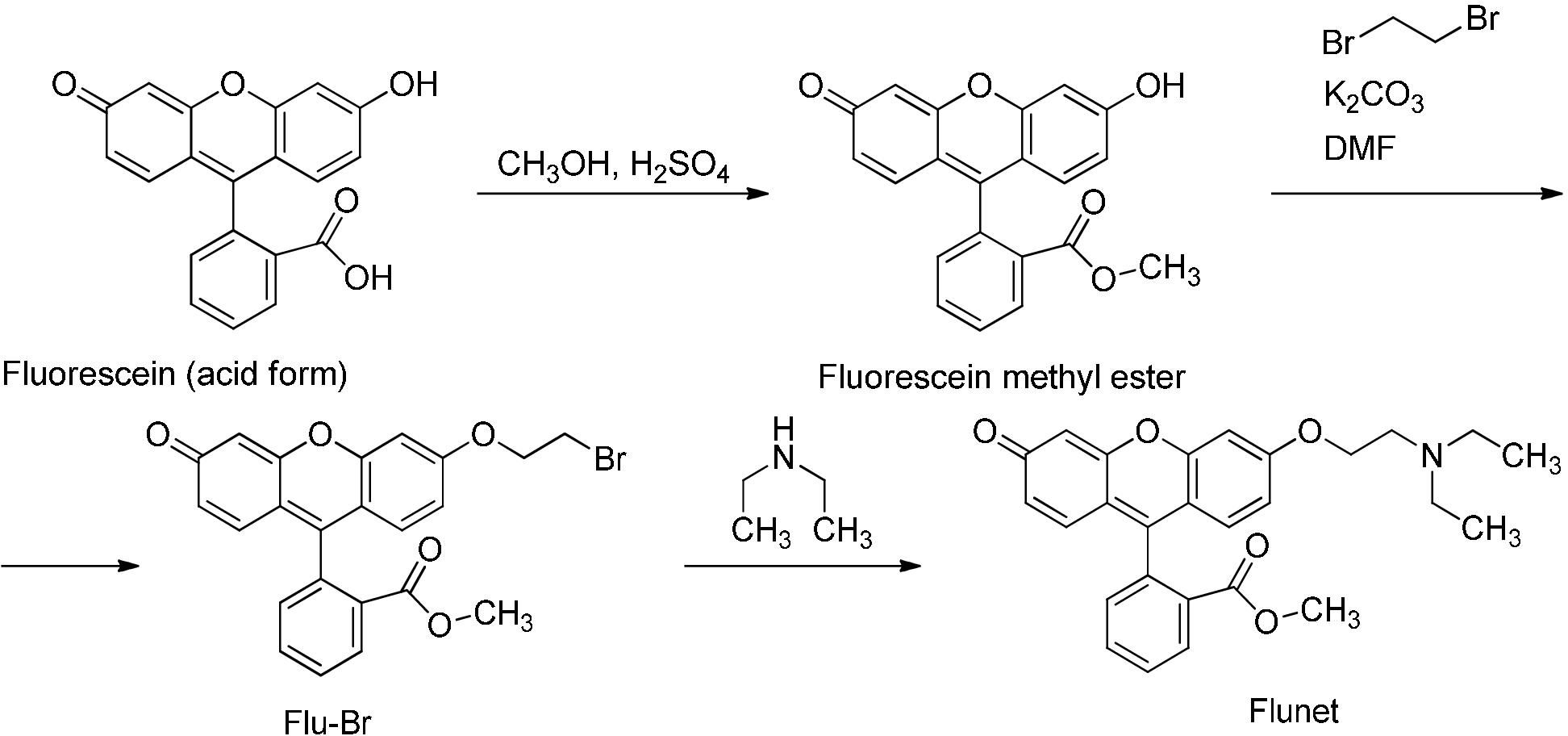
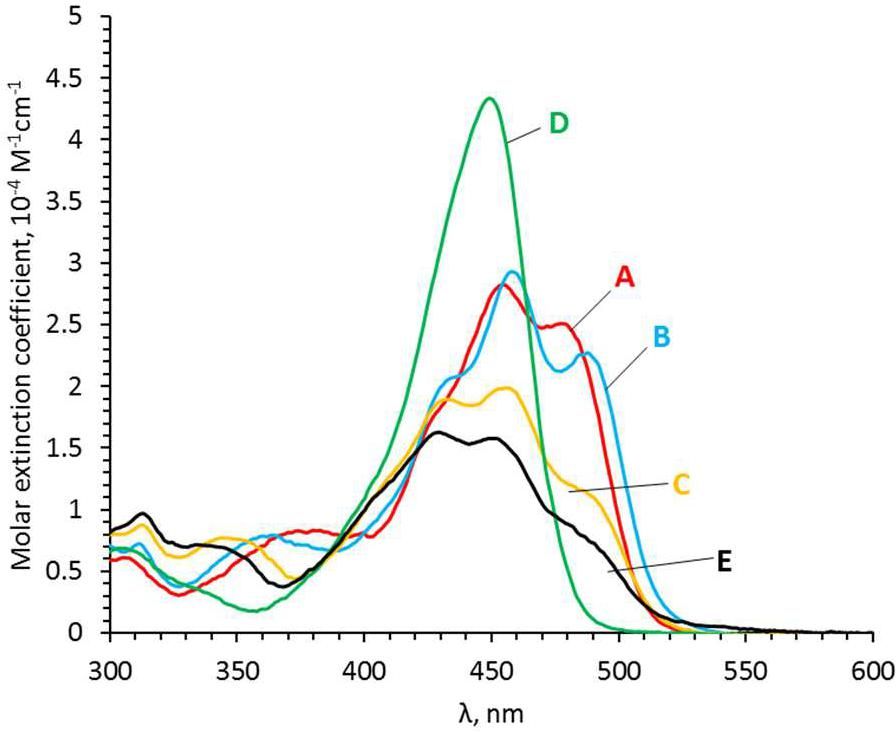
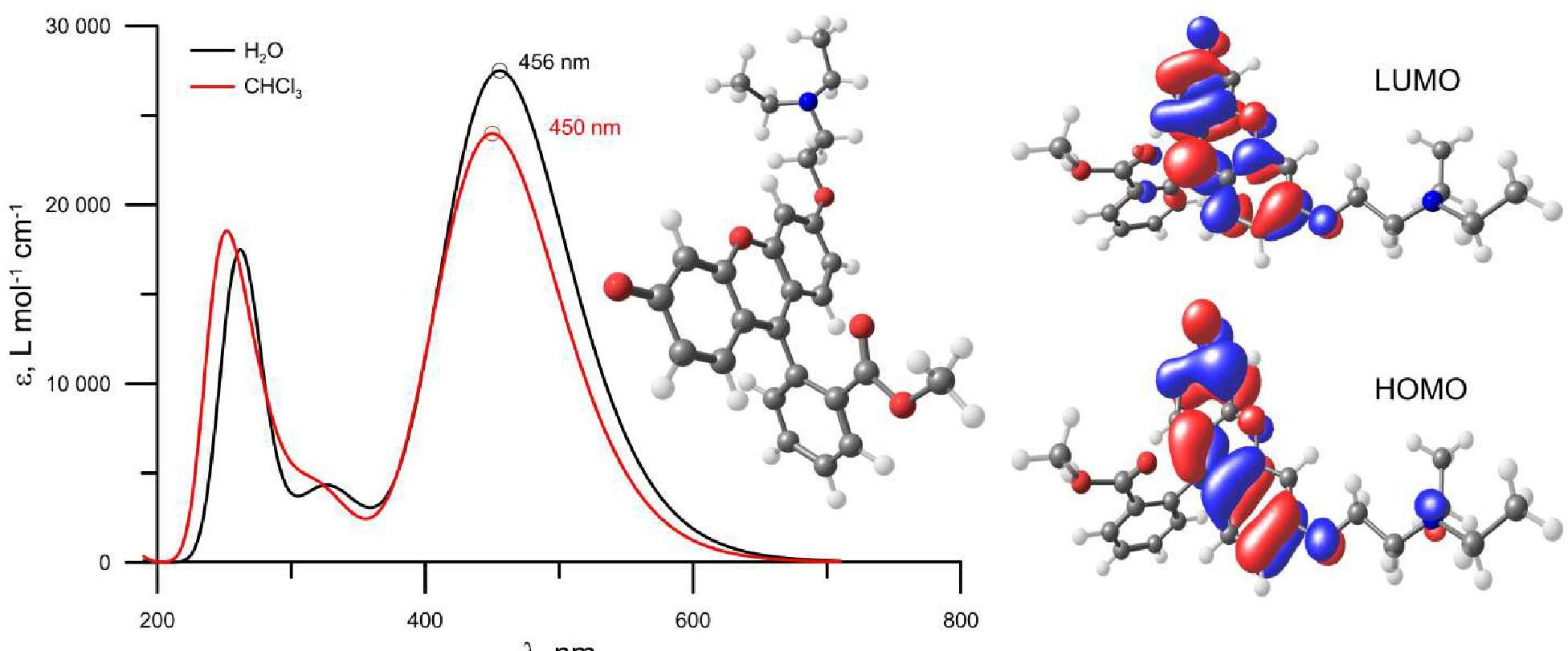
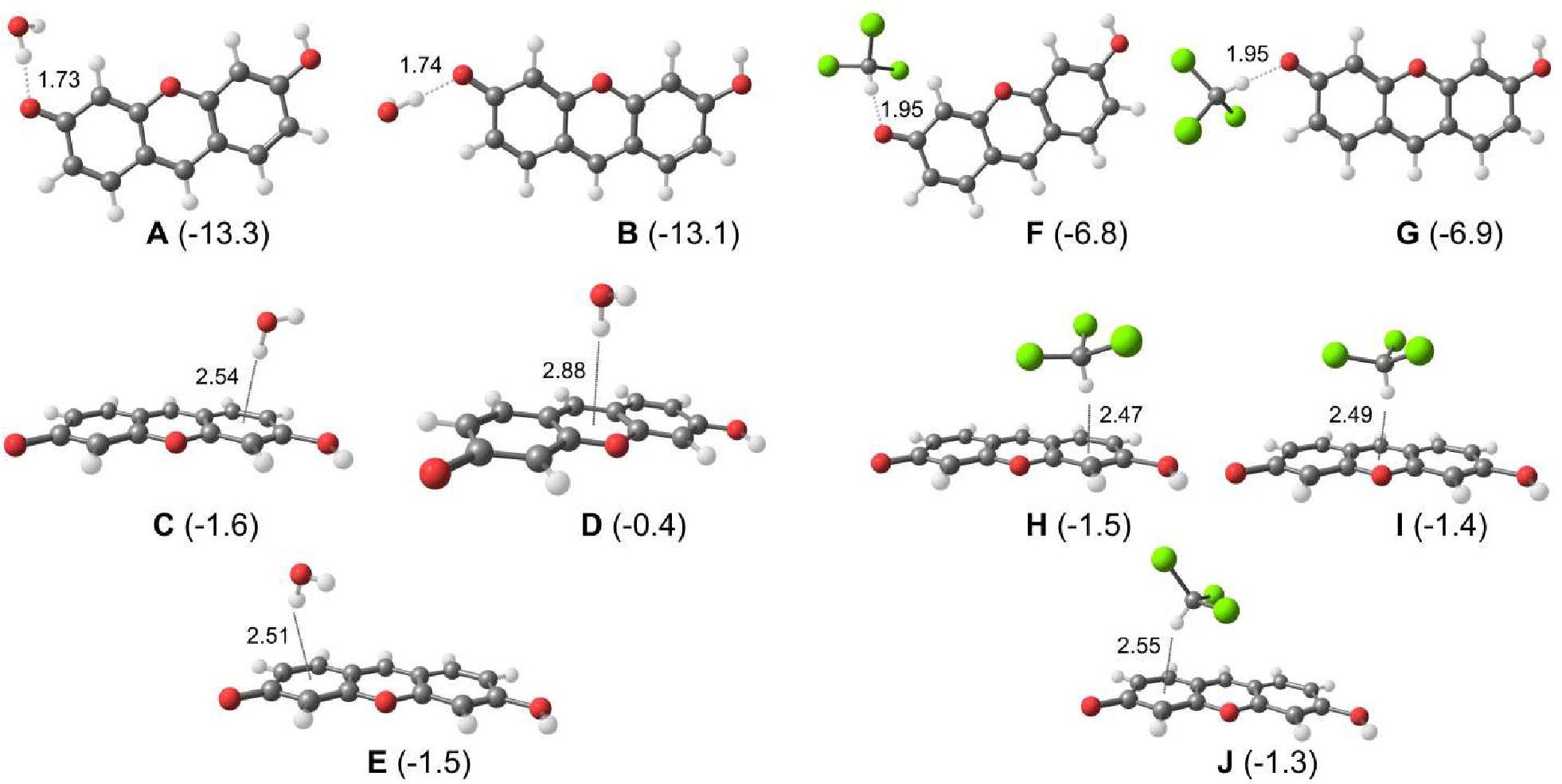
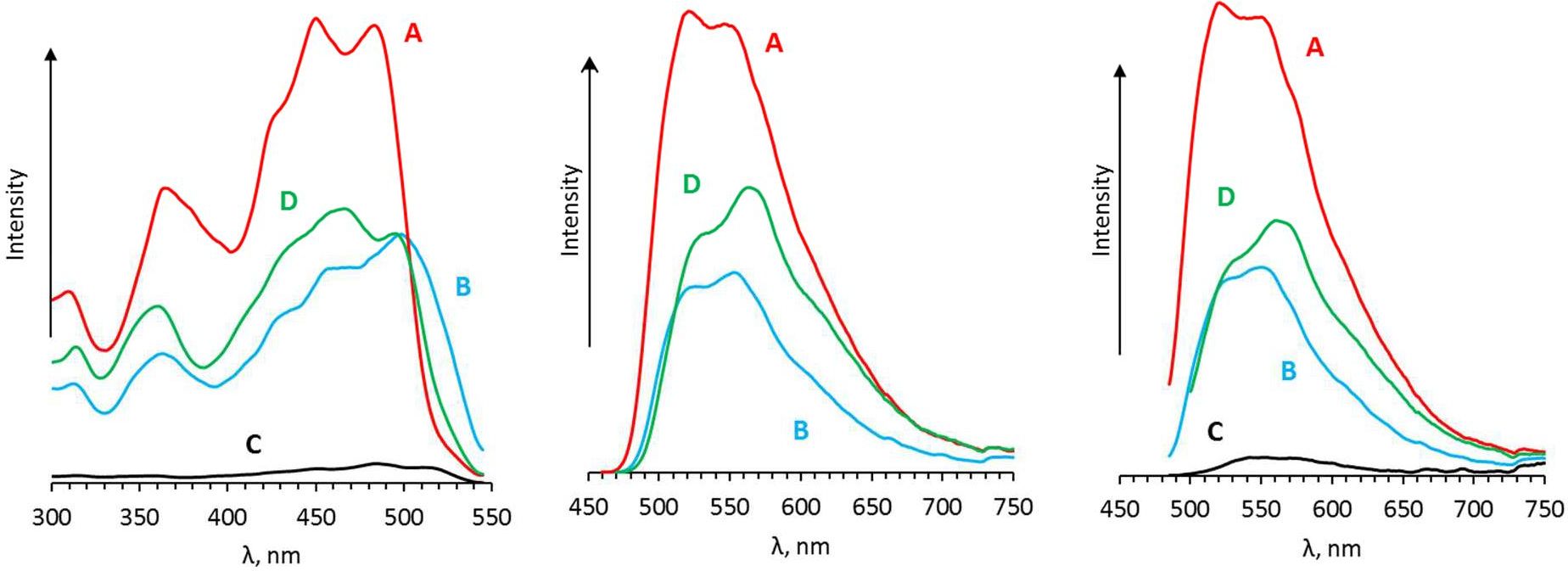
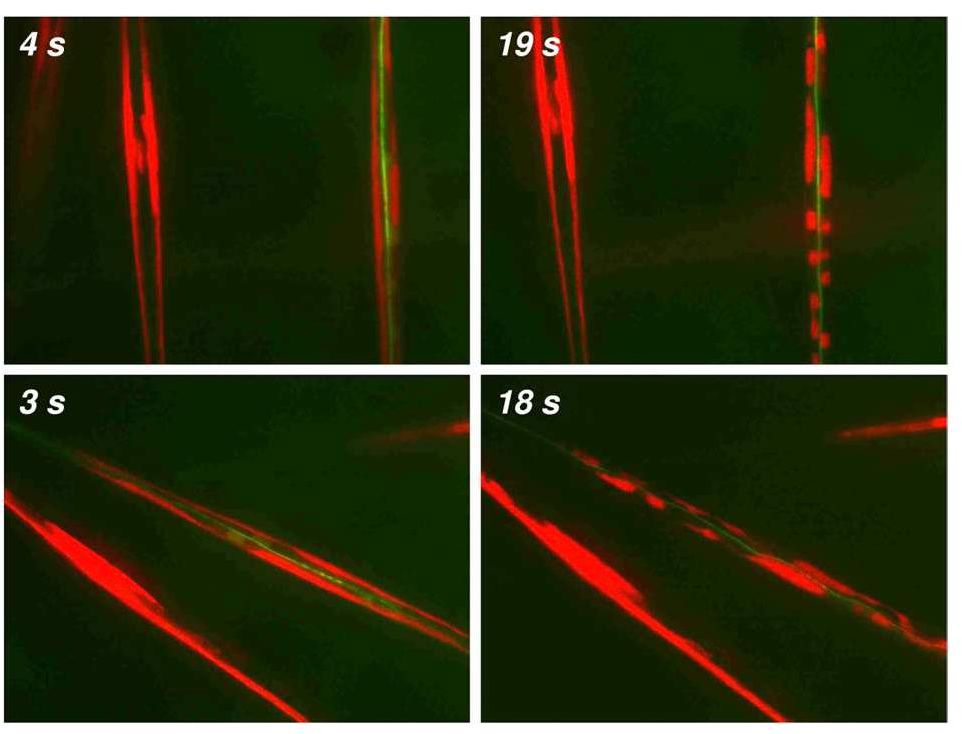
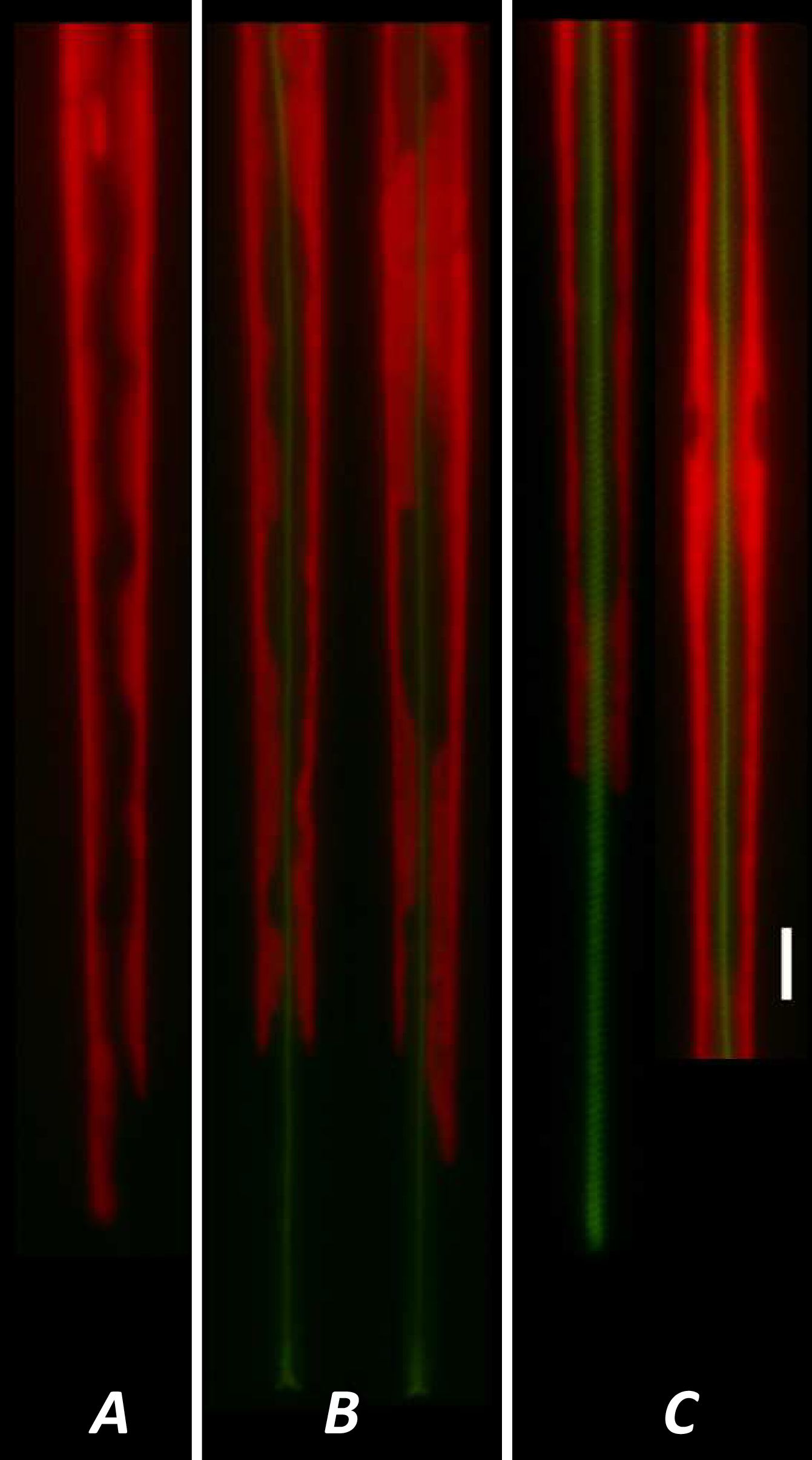
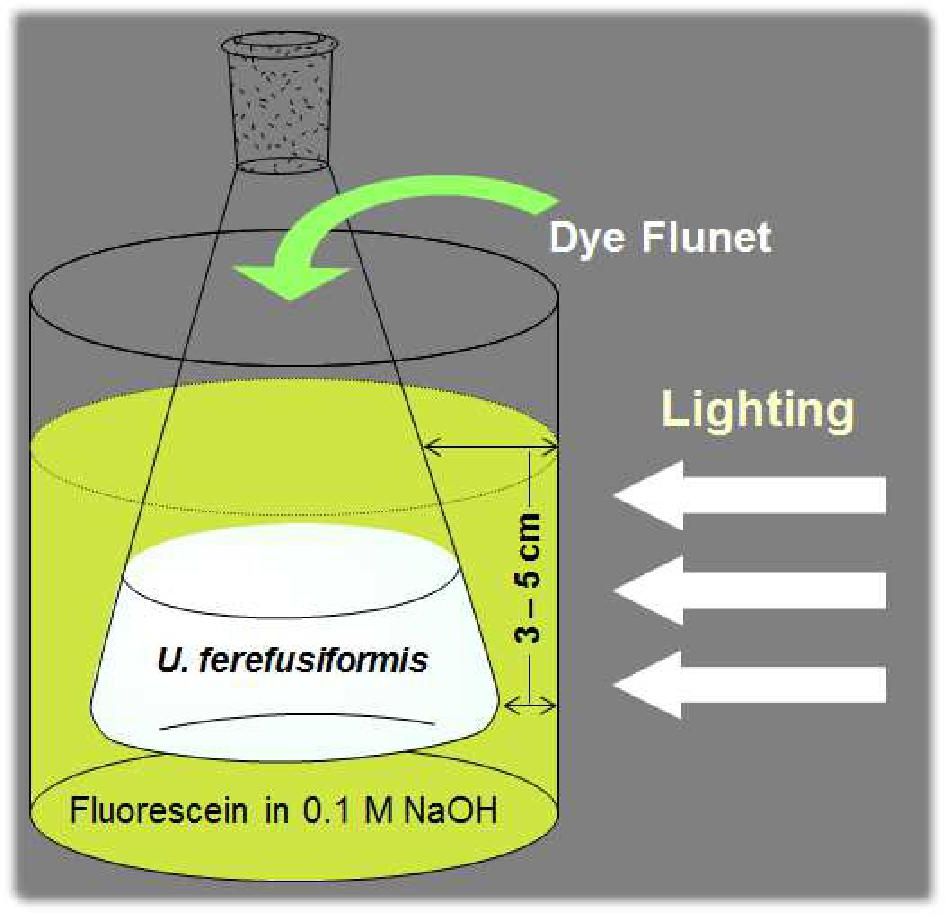
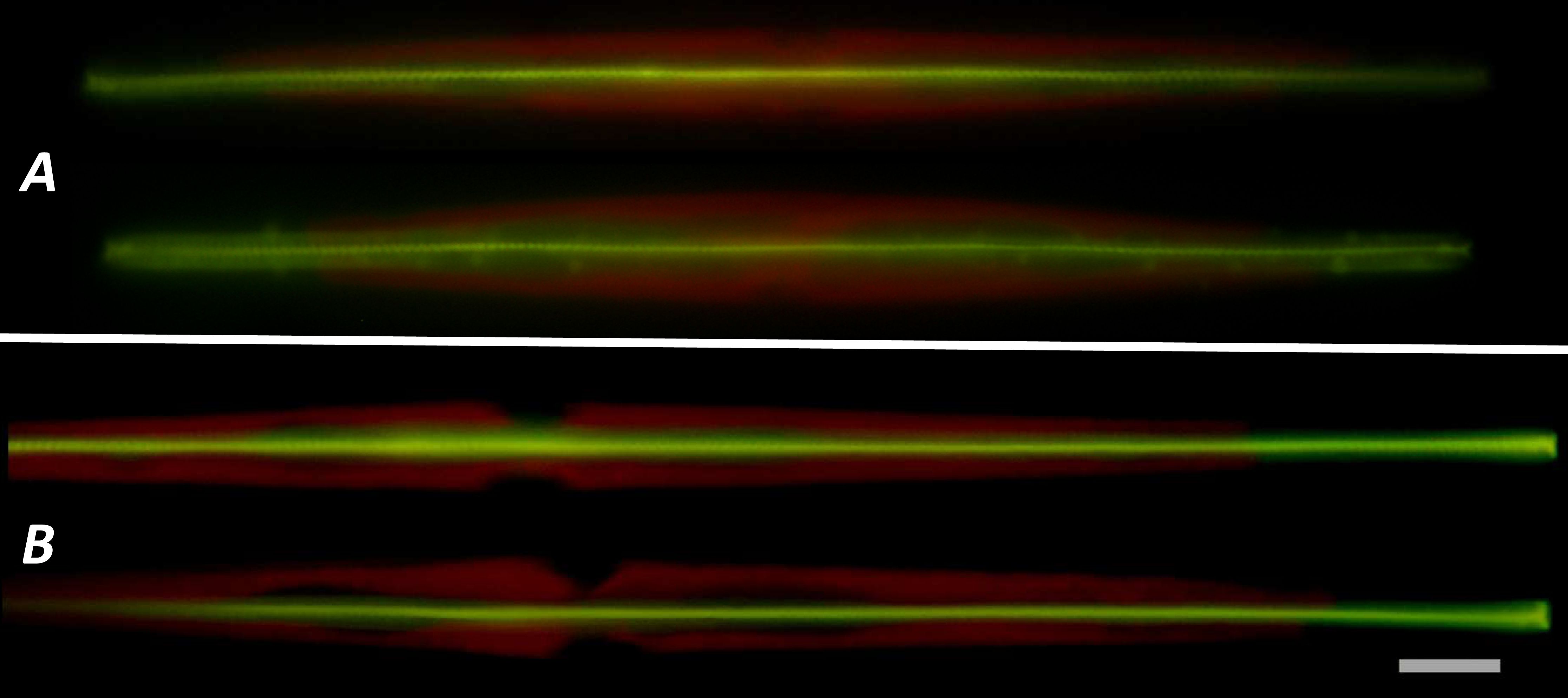
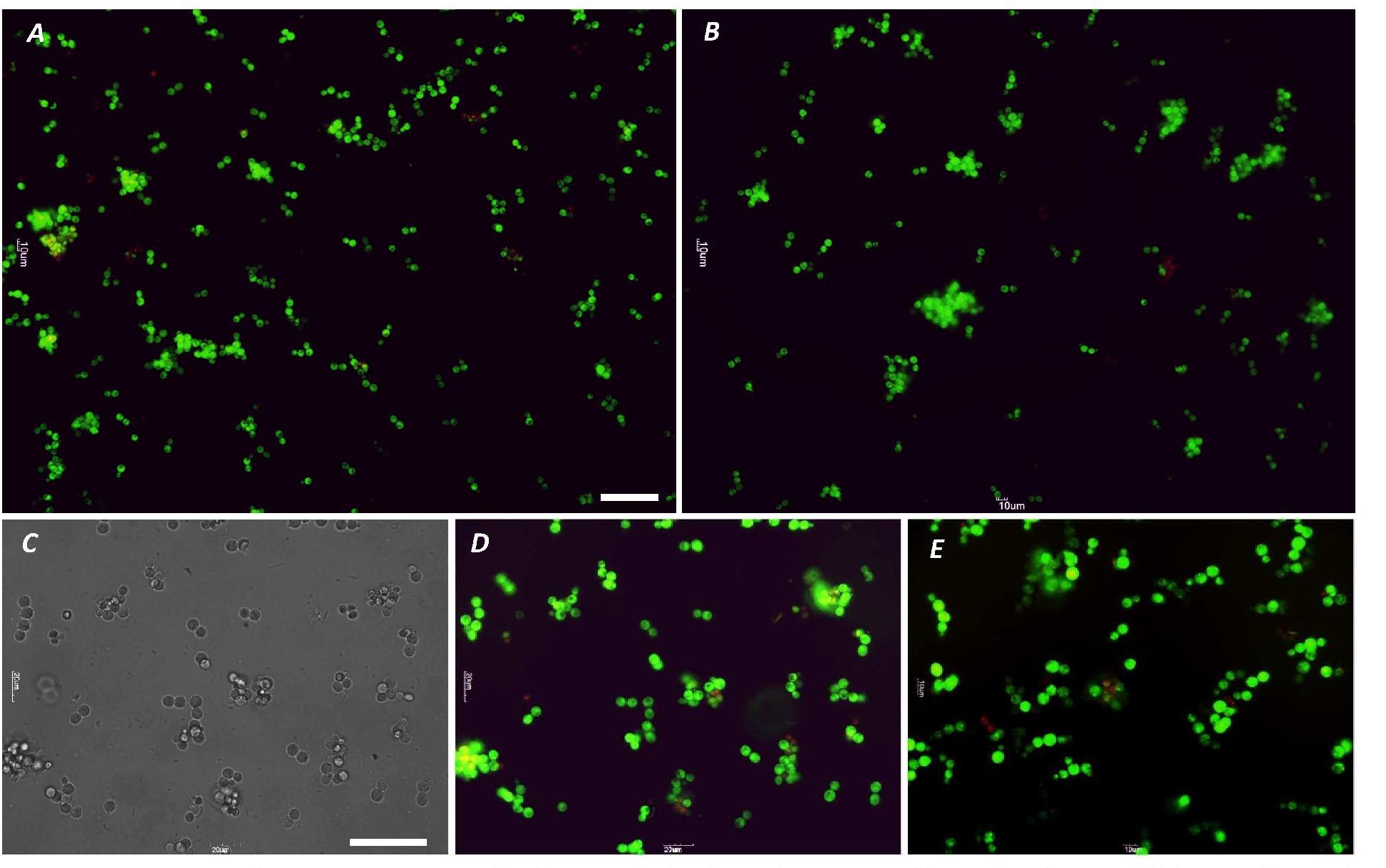
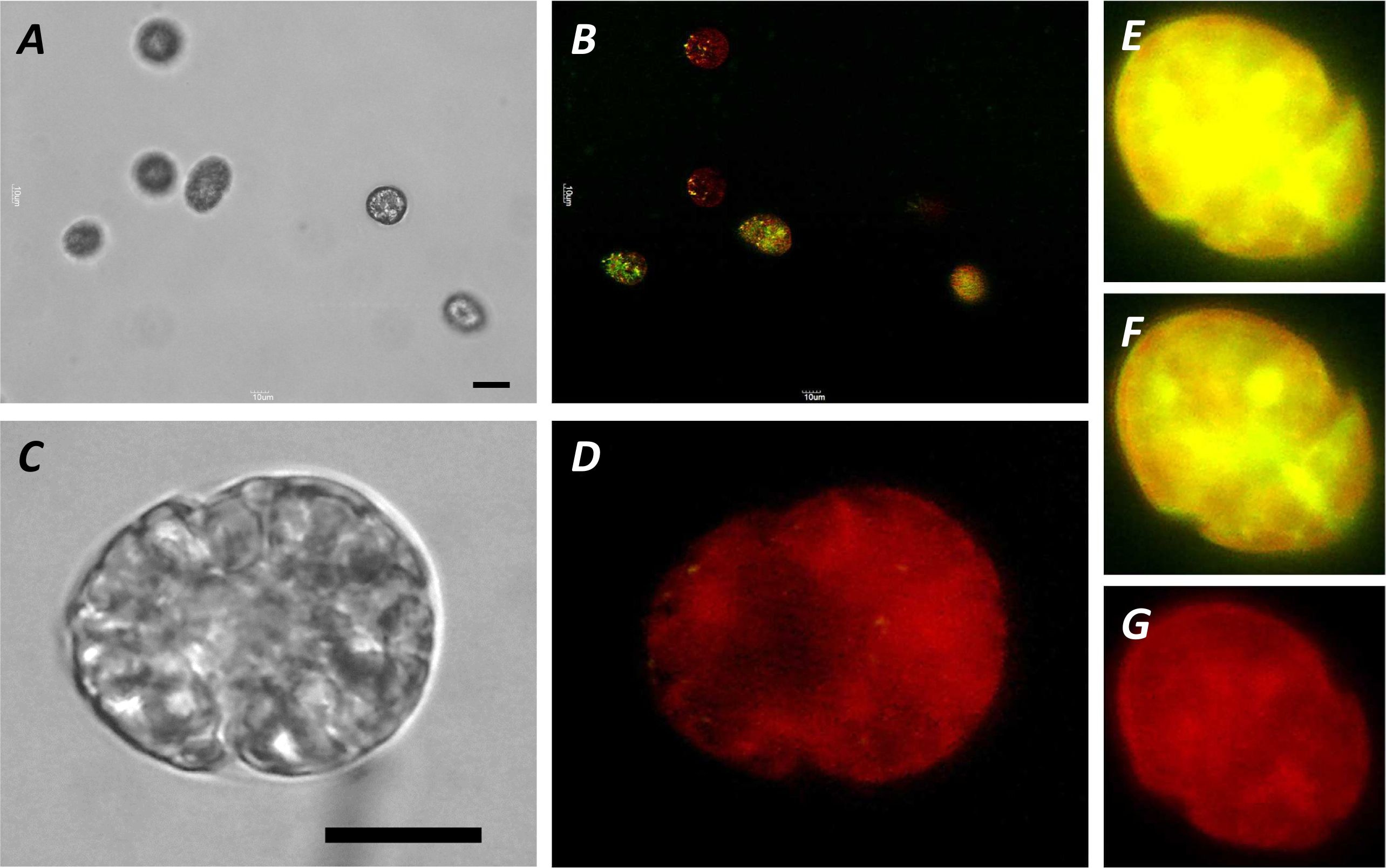
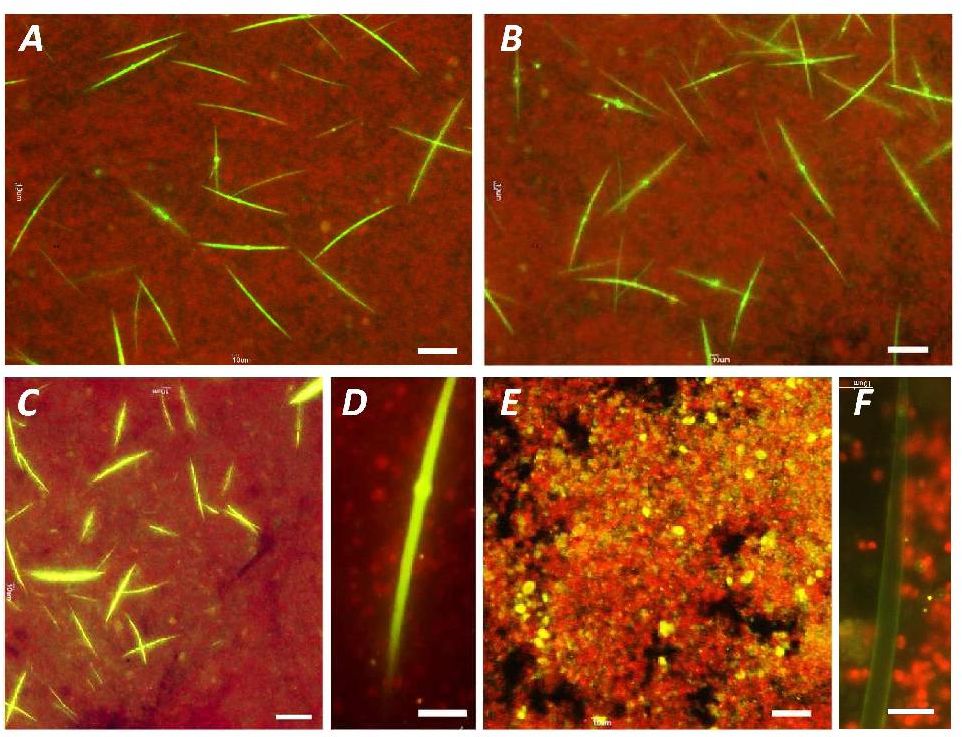
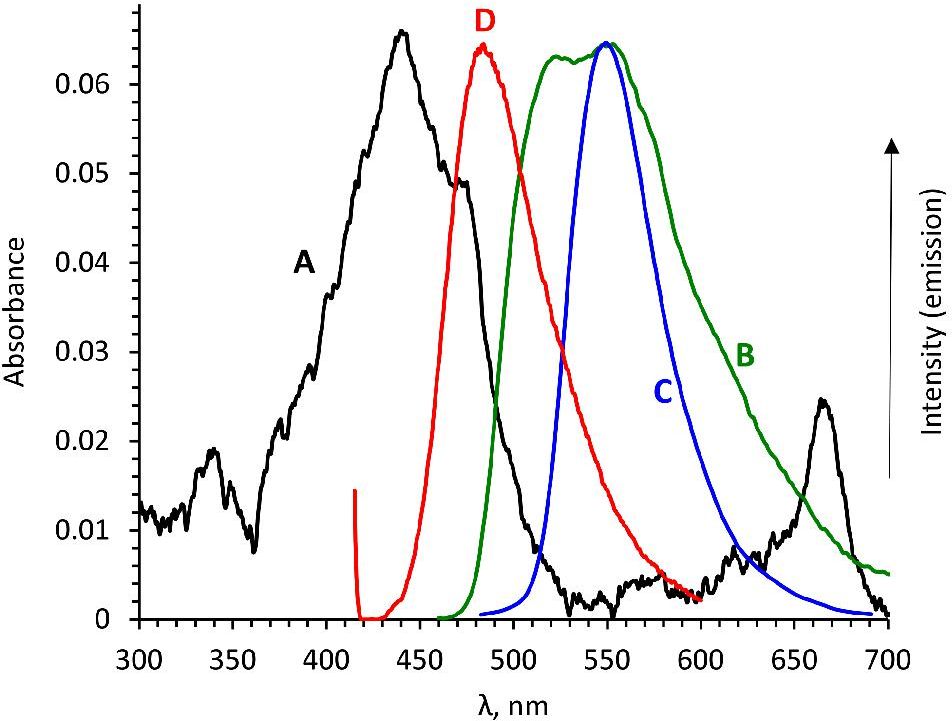
A method for the synthesis of a new amine-containing dye based on fluorescein (Flunet) was developed. The new dye exhibits pH-independent fluorescence in aqueous media and is able to penetrate living cells. Flunet can accumulate in the siliceous valve of diatom algae and cause cell death at a concentration of 0.5 μM, accompanied by chloroplast destruction. Flunet is nontoxic to yeasts and photosynthesizing dinoflagellates, but suppresses the growth of sponge primmorphs. Turning off the fluorescence of Flunet using a fluorescein shield protects diatom algae from death. Flunet, unlike other biosilica vital dyes, has emission in the red region (650-700 nm), which corresponds to the absorption band of chlorophyll. We suppose that once a significant amount of Flunet enters the siliceous valve of a diatom alga, it begins to convert blue light into green and red light, leading to the destruction of chloroplasts located very close (at the micrometer level) to the valve. Siliceous sponges can be suppressed in a similar way: light emitted by growing spicules destroys photosynthesizing sponge symbionts. Our findings open up a new way to influence silica-containing organisms by introducing different fluorescent dyes into the silica structures. This effect can be both positive (increased photosynthesis due to light tuning) and negative, leading to phototoxicity. Considering the main role of diatoms in biofouling of ships and other surfaces, the use of silica-penetrating dyes is a promising environmentally friendly way of antifouling protection.